The diffusion and osmosis They are passive transport processes (they do not require extra energy to occur, as opposed to active transport) of particles of one or more substances that are in different concentrations of solute and come into contact with each other, either through a semi-permeable membrane. (particular case of osmosis) or some other means.
What is broadcasting?
Diffusion is a physical process which is based on the flow of particles of a substance from an area of high concentration of solute to one of low concentration, until the concentration is approximately the same in both areas. The movement of the particles is based on their kinetic energy, that is, there is no external contribution of energy for diffusion to occur. For instance: the passage of oxygen in the pulmonary alveoli.
This movement occurs in any of the states of aggregation of matter, but is more easily observed in the case of liquids and gases. The tendency of the movement is towards the formation of a uniform mixture of the two types of particles.
The scientist Adolf fick established in 1855 some laws that bear his name, and describe various cases of the diffusion of matter in a medium in which initially there is no equilibrium. These laws relate the flux density of the particles to the difference in concentration between the two separate media, its diffusion coefficient and the permeability of the membrane (in case the separating medium is a semi-permeable membrane).
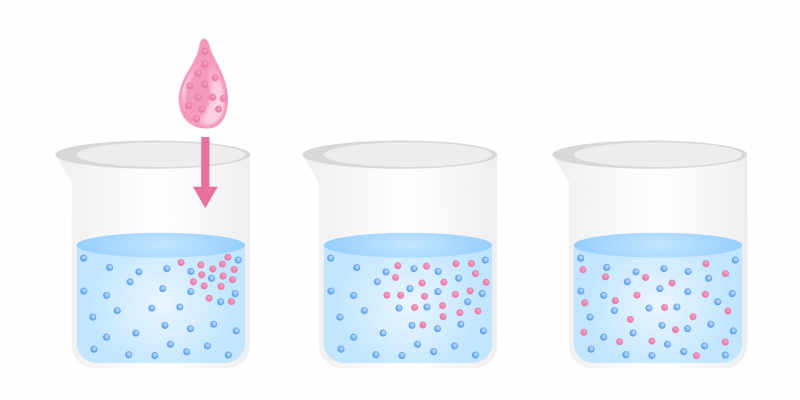
Examples of diffusion
- The passage of oxygen in the pulmonary alveoli.
- Nerve impulses, which involve sodium and potassium ions through the membrane of the axons.
- If a diffuser pair made up of two metals brought into contact across their faces is taken, heated above the melting point and then cooled, it will be verified that the composition has changed: diffusion of the nickel atoms towards the copper occurred and vice versa.
- The warming and color change of a cup of coffee when a good proportion of cold milk is added.
- The entry of glucose to red blood cells, coming from the intestine.
- In an estuary, there is a less dense diffusion of river water that flows over seawater.
- If you put a tablespoon of sugar in a glass of water, the sucrose molecules diffuse through the water.
- The diffusion of gases can be seen when a perfumed person enters a closed place, and everyone immediately senses the smell. The same happens when someone smokes indoors.
What is osmosis?
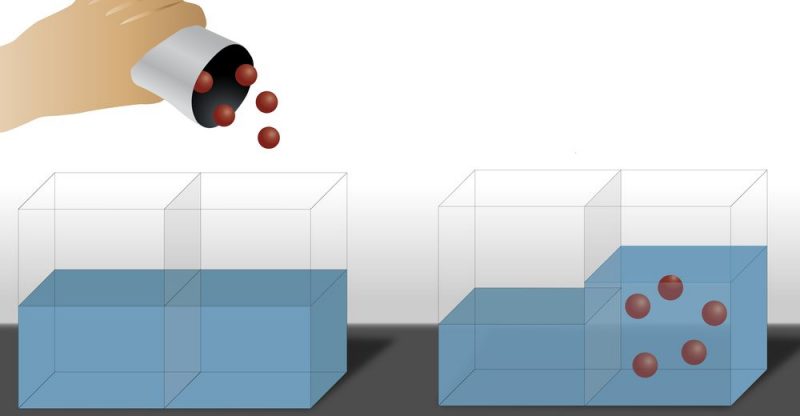
Osmosis constitutes a type of diffusion, but with two particular characteristics:
- Particle flow occurs through a semi-permeable membrane, that is, a porous membrane with pore size at the molecular level.
- Only the solvent particles pass through the membrane.
In this way, it is observed that the solvent tends to pass through the membrane in the direction of the dissolution whose concentration is higher, which ends up causing the amount of solvent to increase in the most concentrated part (in solute) and decrease in the part less concentrated (in solute), that is, the solvent moves from the area with the lowest solute concentration to the area with the highest solute concentration. This is a process that is repeated until the concentration (both solute and solvent) of the solution is equal on both sides of the membrane.
There is also the Inverse osmosis, where the passage of the solvent occurs in the opposite direction than conventional osmosis, that is, the solvent moves from the solution more concentrated in solute to the less concentrated in solute. To achieve this, pressure is applied to the solution more concentrated in solute.
Because it is important?
The solubility of the solute in the solvent and the nature of the semipermeable membrane to be used are the fundamental factors that determine the efficiency of the osmotic process: one of the factors that influences the so-called ‘solubility’ are the interactions between the components of the solution.
The osmotic process It is essential in biological processes where water is the solvent, especially in those processes aimed at maintaining water and electrolyte balance in living beings, regulating water levels in the cell or in the body in general: without this process , there could be fluid regulation and nutrient absorption.
Examples of the osmosis process
- Single-celled living things that live in fresh water enter large amounts of water.
- The absorption of water by the roots in plant organisms, which allows growth.
- Obtaining water from the epithelial cells by the large intestine.
- Split a potato, placing a little sugar with water on one end and a plate with water on the other. The potato acts as a membrane, and after a while it will be seen that the solution that has sugar now has more liquid.
- The hormone ADH that allows the reabsorption of water by the collecting duct in the kidneys.
- The elimination of very dilute urine by which the fish expel the maximum liquid with the minimum loss of salts.
- The elimination of water through sweat in people.
- Filters to purify water work with reverse osmosis, since they are made with a material that allows the passage of water, but not larger molecules.
